 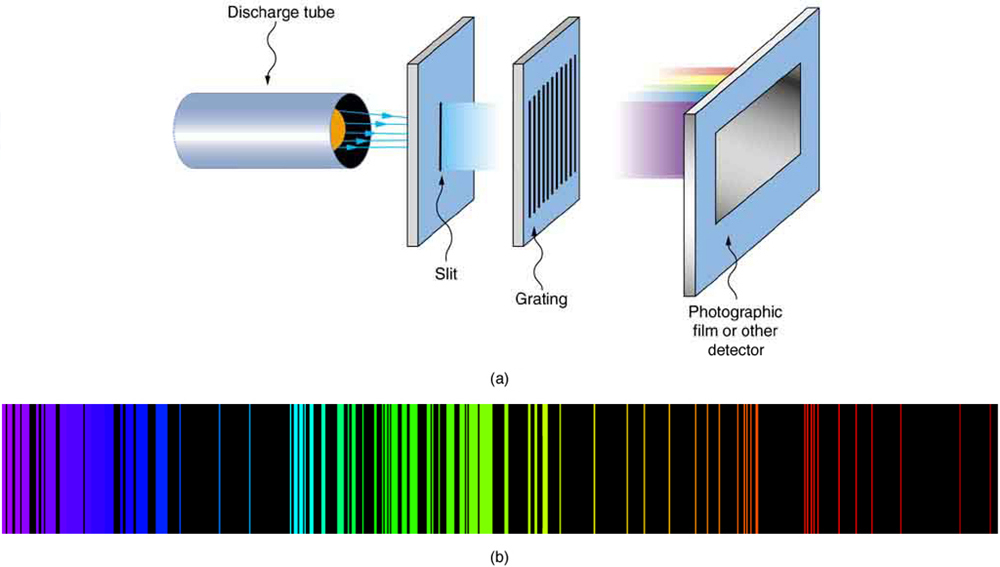
The multiple lines on the spectrum demonstrate the various difference in energies between the ground electronic state and higher energy excited states. This is completely dependent upon the energy of the photon which meets the electron. Upon excitation with a photon, the electron could jump to the next unoccupied orbital, or some which are even higher in energy. Visual Representation of Electronic Energy Levels These all serve as metaphorical “steps” which the electron may briefly exist in, before it eventually falls down to the more stable ground state, releasing a singular photon in the process. Excited electronic levels include higher order s, p, d, and f orbitals which the highest energy valence electron can be excited to fill. Just as a ladder contains multiple rungs, and it is impossible to have a height between two rungs when climbing, electrons can only be stable in higher atomic orbital energies or the ground state, but never in between. Quantized energy levels means that each energy level an electron can be excited to is a discrete value. The reason only small, discrete, wavelengths of light are emitted upon relaxation of an electron is due to the inherent quantization of energy levels within electronic structure. Therefore, atomic spectra are the result of energy changes from valence electrons exclusively. Only the highest energy electron can be excited to an excited state, due to its intrinsic instability. You can view the characteristic atomic spectra for multiple elements from Boston University here. These photon’s wavelengths (which correspond to a specific energy) are what comprise an atomic spectra. Once this highest energy valence electron is excited to a higher electronic energy level (excited state) via a photon, its relaxation back to ground state results in a photon being emitted. Hence, each element has different gaps in energy between its highest occupied electronic energy level and subsequent higher energy levels in unoccupied orbitals. Every element on the periodic table contains a different number of electrons in its neutral state. 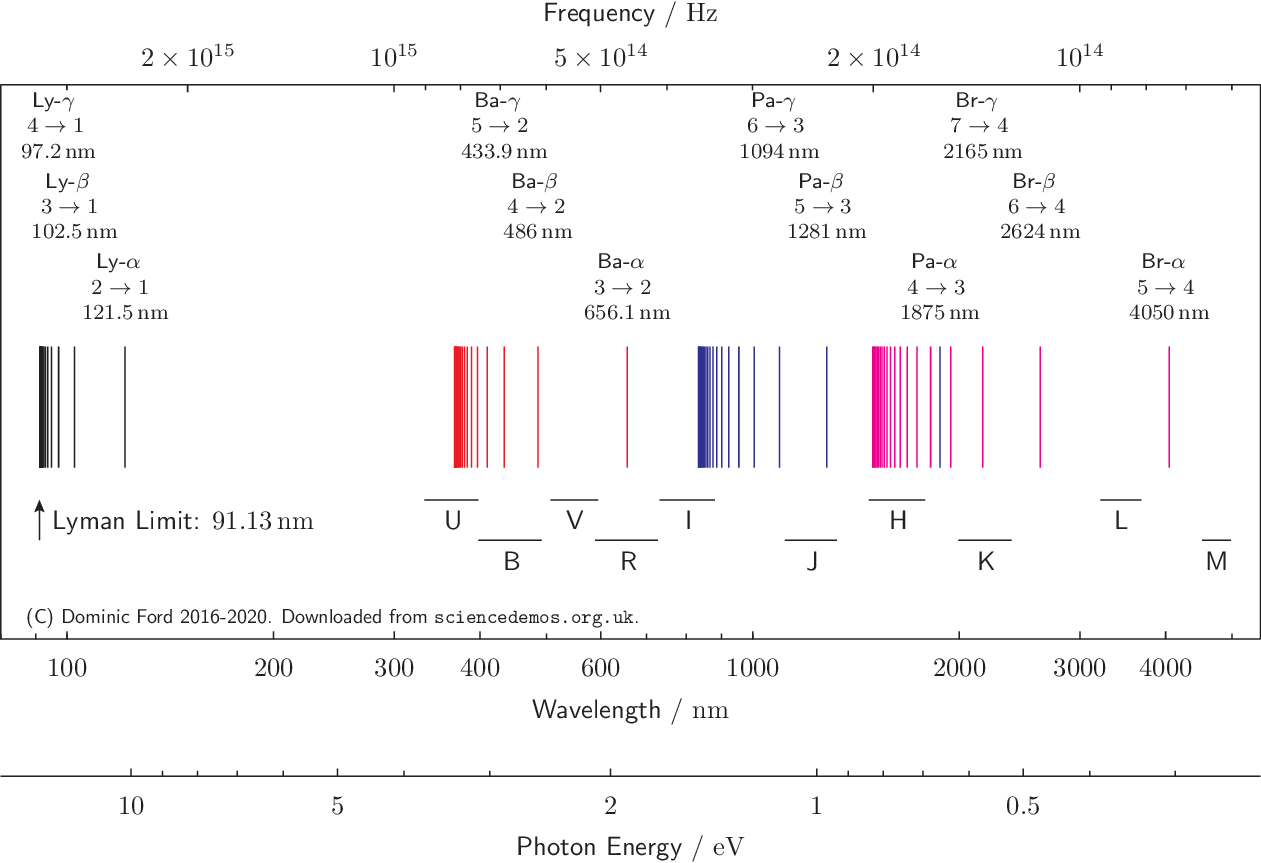 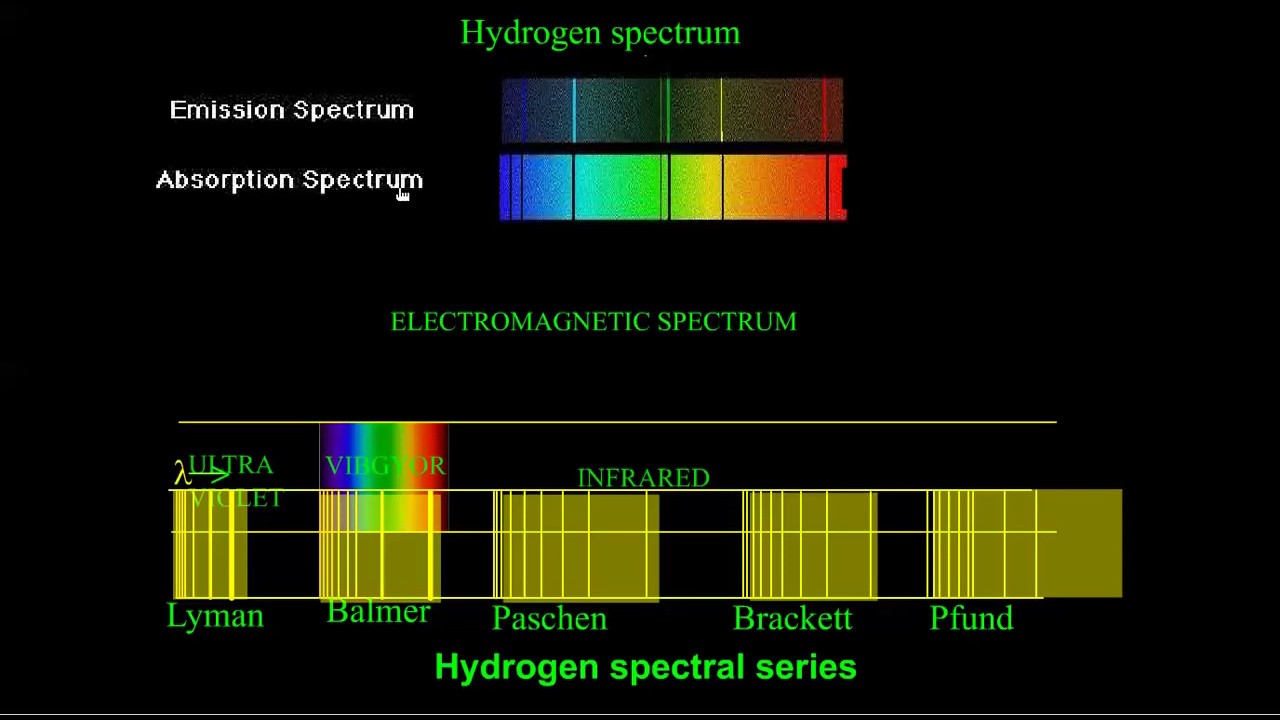
Topics Covered in Other ArticlesĪtomic spectra or atomic emission spectra are the unique wavelengths of light which each element emits due to an electron being relaxed from a higher energy state to a lower energy state in an atom. In this tutorial, you will learn about atomic spectra, including the meaning behind this term, why spectra appear the way they do, and applications of this unique phenomena. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
